Know Your Chemicals
Why it is important to know your Chemicals ?
Chemicals normally referred to as cleaning agents are one of the most critical ingredient to cleaning science as it not only gives Effectiveness and visibility of cleaning standards on the surface but also removes stains.
One of the most important point to be noted while selecting the type of cleaning agent/chemical is how Does it “affect” (damage) the surface on which it is applied.
Cleaning & Sanitation
- Where there is life, there will be dirt, dust, garbage, littering, bacteria, viruses, germs & so on so forth.
- The havoc played by SARS and Bird-Flu viruses should suffice to bear out the importance of sanitation.
Definition & Cleaning Chemicals
- To physically remove : soil, garbage, dust, dirt, grime, grease, stains etc.
- It also causes bacterial reduction through physical removal and is a preliminary step before sanitising / disinfection.
- Any chemical, that aids this process, is called “Cleaning Chemical” or “Cleaning Agent”.
- Soaps, detergents, shampoos, shaving creams, toothpaste, tooth powders, glass cleaners, floor cleaners, toilet cleaners etc. are all cleaning chemicals.
Definition of Germicide
This is a product, which kills germs (bacteria).
At different concentrations, a germicide performs different functions, viz.,
- Sanitising:
To reduce bacteria to an acceptable level from public health standpoint.
This term generally applies to food-service-applications.
- Disinfecting:
To kill all vegetative bacteria, but not the bacterial spores.
Generally applies to health-care, hospitality and environmental cleaning.
- Sterilising:
To kill ALL microbial life forms, including bacterial spores. Generally applies to healthcare equipments and items used for human internal contact, e.g., surgery-items.
A SPORE is a very stable and resistant form of bacteria.
It has thick cell walls, which provide protection against unfavourable conditions, e.g., chemicals and temperature.
A product may function as a sanitizer or as a disinfectant, depending on the concentration. The choice of the correct product is dictated by whether the customer needs sanitising/disinfecting.
Why do we need Chemicals?
- It is well-known that dirt, dust, soiling happens due to human consumption behaviour.
- OIL is one of the basic necessity of human living & consumption. thus dust, dirt, soiling are oily in nature.
- It is also easy to see that water would be the most convenient cleaning medium, because of its extraordinary WETTING POWER AND ALSO BECAUSE OF ITS EASY RINSING ABILITY.
But, water and oils do not mix with each other.
- Thus, water, by itself, cannot effect cleaning, though it can be a cleaning medium.
What are Surfactants?
To overcome this, chemicals have been synthesised, which have both:
- A water-loving component and
- An oil-loving component.
Both these components are present in every chemical. These chemicals bring about a union at the surfaces of water and oil and make them miscible with each other. Therefore, these are known as surfactants, i.e., they have “surfaceactive” properties.
They act as a bridge between the oils and the water, because a surfactant mixes well both with water and oil. They also further enhance the wetting power of the water by reducing / eliminating the repulsion between oils and water. Because a surfactant is miscible with water, a solution of the surfactant can be formed in water. When this solution comes into contact with oily soils, the surfactant inside the solution mixes well with the oily impurities too.
How Surfactants AID cleaning?
- The soil has a greater affinity (attraction) for the surfactant than it has for the surface it has made dirty.
- Therefore, it leaves that dirty surface and gets attached to the surfactant and through the surfactant; it gets attached to the water-based solution.
- Since the soil has been loosened from the dirty surface and has entered the water-based solution, the entire tri-partite mixture i.e. oily impurities + surfactant + water, can be rinsed away using pure water.
- Scrubbing / any mechanical action like brushing, abrasion, can further enhance the loosening of the soil from the surface.
- Surfactants are the most basic and the most important ingredients in a cleaning chemical.
Addtives
Depending upon the requirement, many additives are included e.g.
- Disinfectants
- De-odorising agents such as perfumes
- Anti-corrosion agents
- Hard water softeners
- Anti-re-depositing agents and many, many others
Depending upon the type of surfactant used and the additives, various types of cleaning chemicals can be synthesised for a wide array of applications, e.g.
- Soaps
- Detergents
- Shampoos
- Tooth-pastes, Tooth-powders
- Glass-cleaners
- Floor-cleaners
- Toilet-cleaners
Various types of Surfactants
1. Anionic surfactants: are those, in which, the active ingredient, i.e., the basic surfactant species, is an anion, i.e., a NEGATIVELY charged ion. types of anionic surfactants: –
- Sodium stearate
- Sodium lauryl sulphate
- Sodium lauryl ether sulphate
- Alpha-olefin sulphate
- LABSA sodium salt or
- sodium linear alkyl benzene sulphonate
Most bathing soaps, shampoos, shaving creams fabric detergents, tooth-pastes and tooth-powders contain anionicsurfactants. Anionic surfactants are very versatile and are suitable for a wide range of surfaces, e.g., glass, metals, plastics, ceramics, porcelain, granite, marble, Rexene, etc. They are also relatively cheap, very easy to manufacture on a mass scale, because of the abundant availability of the raw materials and are therefore the most widely used type.
2. Cationic surfactants are those, in which, the active ingredient, i.e., the basic surfactant species, is a Cation, i.e., a POSITIVELY charged ion.
- Quaternary ammonium-based cationic surfactants are quite common. These are very effective disinfecting chemicals.
- Fatty amine ethoxylates are another type of cationic surfactants found mostly in premium quality toilet cleaners.
Thus, they are used for special applications.
3. Non-ionic surfactants do not contain any ionic chemicals.
- Fatty alcohol ethoxylates are popular surfactants.
- These are also very versatile and are suitable for a wide range of surfaces, e.g., glass, metals, plastics, ceramics, porcelain, granite, marble etc.
- They are expensive and used for high quality, high-end, specialised applications, e.g., emulsification, i.e., removal of very greasy, highly oily impurities.
- Premium quality industrial laundry detergents for textiles also contain non-ionic surfactants.
- Similarly, for cleaning delicate and expensive surfaces like marble, these can be effectively used.
4. Amphoteric surfactants:
- Contain both anionic & cationic surfactants.
- So arranged so that they don’t interfere with each other.
- Both work independently.
- Anionic is busy in cleaning. Cationic is busy in disinfection generally.
- When they get in touch with alkali, they behave like acid to make neutralizing effect and vice versa.
Formulation of A Chemical
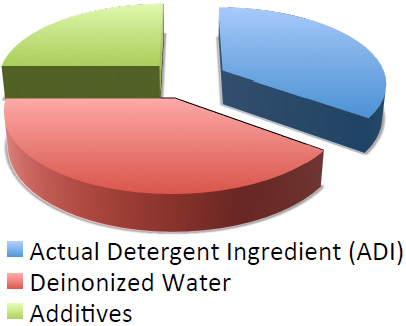 Surfactants, the soul of chemicals
Surfactants, the soul of chemicals
Nature of Soiling
- Everyday soiling:
- Soils get stuck to the surface through an oily medium. Oils are acidic nature. (They are organic compounds).
- Soils stick to the rail coach surface through moisture too. The pH of the moisture (humidity) in air is typically in the acid range: 5.5 to 6.5.
This is because it contains small quantities of dissolved gases like carbon-do-oxide CO2, oxides of nitrogen, sulphur-di-oxide SO2. All these are acidic gases.
- Likewise, ambient air is also acidic, because ambient air too contains these aforementioned gases. Rail coach surfaces might also contain entrapped air bubbles between the oily medium, soils
and moisture.
All these make the total soil mass acidic.
- Special types of soils, e.g., grease lubricating oils.
They are inherently acidic in nature. Therefore, all detergents contain added alkali in some form. The alkalinity neutralizes acids and aids soil removal.
Grease-removers are especially strongly alkaline, containing NaOH and KOH, potently strong alkalis. Caustic soda or sodium hydroxide, caustic potash or potassium hydroxide.
Ph & Its effect on various Surfaces
Most soils are generally acidic in nature, e.g.:
- Body sweat
- Body oils
- Fabric soils
- dirt and dust on surfaces like glass, metals, floors, bathroom fixtures and chrome fittings etc.
- Therefore, commonly used cleaning chemicals are alkaline in nature to neutralise this acidity.
- This alkali chemically reacts with the acidic soils to create new, water-miscible entities, which can be rinsed away in running water.
- This chemical reaction is called saponification. Thus, the alkalis supplement, enhance and accelerate the cleaning action of the surfactants.
- Alkalinity is brought about by adding chemicals like sodium carbonate (soda ash, washing soda), small quantities of caustic soda, etc.
- The pH of such chemicals is typically between 7.5 to 9.5, depending on the degree of soiling.
- Heavy soils like grease require higher alkalinity, i.e., pH in the range of 11 to 12.
- As a general rule, higher the degree of soiling, higher the alkalinity of the chemicals.
- However, strongly alkaline chemicals should not be used on aluminium, zinc and painted surfaces, as the strong alkali can damage these materials.
- Human fasces dissolve in acidic medium and can be rinsed away. Therefore, toilet-cleaners are acidic in nature.
- Their pH is in the range 2 to 5.
- While these chemicals are suitable for ceramic and porcelain, they will attack and damage metallic toilets used in Railway coaches in India.
- For such toilets, 3rd category must be preferred.
Ph Scale

pH neutral chemicals are neither acidic nor alkaline. They have a pH of 7.
Chemistry Types
| Aggressive Chemistry |
Sophisticated Chemistry |
- Shows strong visible efficacy
- Strong Alkali based constituents or strong acidic
- Eats away the gloss/texture/ shine/colour of the surface in the long run
- Bad for health & environment
- Requires thorough rinsing hence more water
- 80% industrial users choose this type of chemicals
|
- Shows efficacy as required
- Mild alkali or acidic constituents
- Does not have any such observations in the long run
- Safe for health & environment
- Gets rinsed easily
- Only 20% industrial users choose this type of chemicals
|
Dilution Ratios & Time Effect
- Considering the ease of transportation, economy and convenience of handling and usage, cleaning chemicals are often made in concentrated form, with little or no water being added.
- Depending on the use and the nature and degree of soiling, these concentrates may be diluted with the addition of water to make it “Ready-To-Use” (RTU).
- Cleaning efficiency increases with time up to a certain period, beyond which, it stabilises and there is not further cleaning action, even if the action is continued for a longer time.
Chemicals & The Environment
- All these synthetic chemicals can be pollutants for the soil and for the water bodies. Therefore, discharge as effluent must be done carefully.
- The surfactants should be bio-degradable, i.e., commonly found bacteria in the environment should be able to break down the surfactants into small, harmless chemicals, e.g.:
- Water
- Carbon-di-oxide
- Simple inorganic salts like sodium sulphate etc.
which can be safely discharged into the soil or water bodies or a waste site.
From eco-considerations, the cleaning chemicals MUST have the following attributes:
- No phenol-based surfactant, viz., APEOs, alkyl phenol ethoxylates. (octyl phenol ethoxylates nonyl phenol ethoxylates). Banned constituents.
- No organic solvents like butoxy ethanol, xylene, benzene, carbon-tetra-chloride, methanol.
- No polluting phosphates / phosphonates.
- No formaldehyde and harmful carcinogenic ingredients.
- No EDTA-based products. (Ethylene diamine tetra acetic acid).
- No NTA (Nitrilo-Tri-Acetic Acid) or its sodium salt.
- No ammonia, no strong acids.
- No aerosol-based chemicals in perfumes or sprays. Aerosols can harm the atmosphere and the ozone layer.
- The USEPA – United States Environmental Protection Agency is a respected regulatory certifying body.
- A product registered with EPA is considered a safe cleaning chemical.
- The Governments of developed countries have formulated well-defined and stringent laws and legislation on the all round quality and safety of cleaning chemicals, which are
implemented very strictly.
- Safety to human health, safety in handling and storage and safety to the environment, air, soil, water-bodies.
- Unfortunately, in India, there are no well-defined, stringent laws, rules-regulations and legislation on the safety of cleaning chemicals.
The level of education and awareness of cleaning chemicals among Indian buyers is very low.
Indian Manufacturers may claim their chemicals to be ecofriendly, though it may not be true. Why ??
- The surfactants used may not be adequately biodegradable.
- It is generally considered to be a norm that the biodegradability should be at least 80 to 90 %.
Aggressive Cleaning Chemicals Manufacturers in India may be :-
- Either highly alkaline or highly acidic
- Unsafe for many surfaces
- Unsafe for the environment and
- Unsafe for handling by human being.
These chemicals may contain one or more of the harmful ingredients mentioned above.
MSDS & Its Importance
Considering the environmental issues just discussed, the Governments have made it mandatory for all the manufacturers of cleaning chemicals to furnish
“MSDS – Material Safety Data Sheet”
for the cleaning chemical being supplied.
- It is compulsory to publicly display the MSDS of all the Cleaning chemicals being used in the premises.
- MSDS must contain 16 standard points in accordance with 89/542/EEC.
- The MSDS should be easily accessible and legible.
- Each person working the premises must be given formal, comprehensive training on how to understand the information contained in the MSDS and how make use of that information.
Essential of Hard Surface Cleaning Chemicals
- Must have a properly written MSDS (Material Safety Data Sheet), containing 16 standard points in accordance with 89/542/EEC.
- Must have properly written PIS (Product Information Sheet), with detailed information on usage-procedures, (directions for use).
- Should have active detergent ingredient in the range of 40 %.
- Should be free from alkyl phenol ethoxylates, APEOs., formaldehyde, butoxy ethanol, phosphates or phosphonates
- Should have least 80 – 90 % biodegradable, according to EU
- Should be low to medium in foaming.
- pH to be in the range of 8 to 9.5.
- Should NOT be inflammable
- Should be miscible / dispersible with water in all proportions.
- Should be able to perform well in water with hardness of 150 to 200 PPM and TDS of up to 500 PPM.
- Should not leave behind any residual precipitates with hard water.
- Should not attack emulsion and water-based paints.
- Should contain corrosion-inhibitors.
- Should have storage stability - shelf-life of at least 12 months.
- Viscosity and consistency should not decrease with time and with variations of temperature due to different weather and climate conditions.
Banned Constituents of Chemicals Worldwide
- Alkyl Phenol Ethoxylates
- Mercury & Mercury Sauts
- 2 - Butoxyethenol
- Cadmium or Cadmium Salts
- Choroform
- Chromium & Chromium Salts
- Cresols
- Phenols
- Sodium Nitrolotriacotic Acid
What is An Ideal Chemical?
- No phenol-based detergents, viz., APEOs, which are banned worldwide.
- NO organic solvents like butoxy ethanol, xylene, benzene, carbon-tetra-chloride, methanol.
- No polluting phosphates / phosphonates.
- No formaldehyde and harmful carcinogenic ingredients.
- NO EDTA (Ethylene Di amine Tetra Acetic Acid) -based products.
- NO NTA (Nitrilo-Tri-Acetic Acid) or its sodium salt.
- Ingredients safe to human health and to the environment.
- No ammonia, no strong acids.
- No aerosol-based chemicals in perfumes or sprays. Aerosols can harm the atmosphere.
|

|





